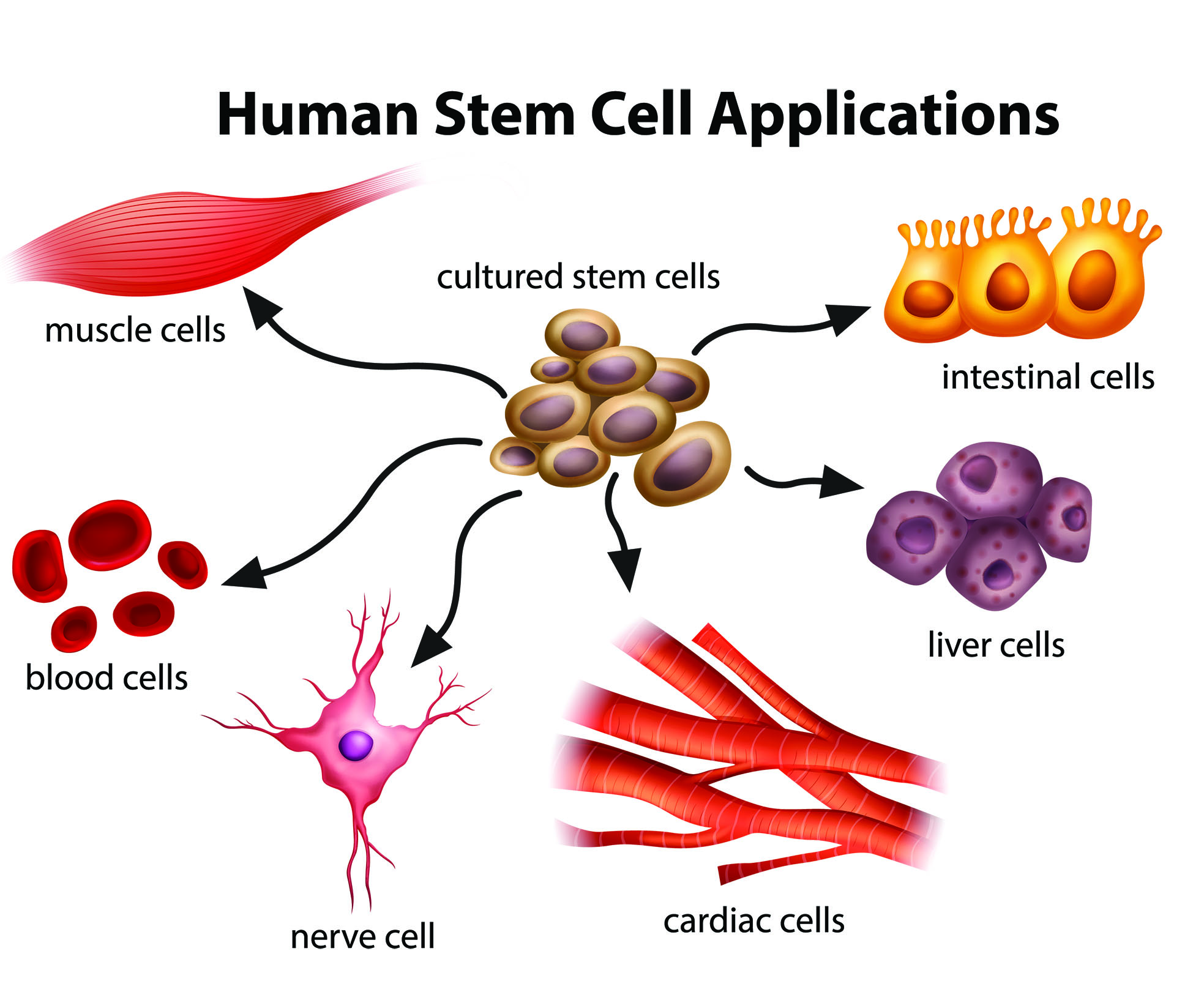
You’ve probably seen them. Those glowing, neon-colored blobs that look like something out of a sci-fi movie or a psychedelic screensaver. They’re everywhere in science news. But honestly, most photos of stem cells are deeply misunderstood by the general public. We see a cluster of bright green circles and think, "Cool, science." In reality, what you’re looking at is a frozen moment of biological potential that has been dyed, stained, and blasted with lasers just so our human eyes can make sense of it.
Stem cells don't actually look like glowing emeralds.
Left to their own devices under a standard light microscope, they are mostly translucent and, frankly, a bit boring. They look like grainy pebbles or stretched-out bits of gum. The magic happens when researchers use techniques like immunofluorescence. They apply antibodies that bind to specific proteins, and those antibodies are attached to "fluorophores." When you hit them with specific wavelengths of light, they glow. That’s why you see those striking contrasts—blue for the nucleus, green for the cytoskeleton, red for specific markers of "pluripotency."
It’s art. But it’s also data.
The Reality Behind the Most Famous Photos of Stem Cells
If you search for these images, you’ll likely stumble across shots of Human Embryonic Stem Cells (hESCs). These are the "O.G." images that fueled the massive ethical and scientific debates of the early 2000s. These photos usually show "colonies." Stem cells are social. They like to huddle together in these circular islands on a petri dish. If they wander off alone, they often die or start changing into something else.
Take the work of Dr. James Thomson at the University of Wisconsin-Madison. Back in 1998, when he first isolated these cells, the photos weren't high-def masterpieces. They were grainy, black-and-white proofs of concept. Today, we have 4K resolution imaging that can show the tiny bridges—nanotubes—that cells use to talk to each other.
Then there are the Induced Pluripotent Stem Cells (iPSCs). This is the Nobel Prize-winning stuff from Shinya Yamanaka. In these photos, you’re looking at regular skin cells that have been "tricked" back into an embryonic-like state. Visually, iPSC colonies look remarkably similar to embryonic ones. That’s the point. The photo is the proof that the clock has been reset.
Why Some Stem Cell Images Look Like Starry Nights
Have you ever noticed how some photos of stem cells look like a Van Gogh painting? That’s usually a "Brainbow" or a similar lineage-tracing technique. In the early 2000s, researchers at Harvard developed a way to genetically modify cells so they express different ratios of red, green, and blue fluorescent proteins.
As these stem cells divide and differentiate, their "children" inherit the color.
Imagine a single stem cell in a developing mouse brain. It’s colored bright yellow. All the neurons it creates will also be yellow. The cell next to it might be teal. This creates a map. When you look at a photo of a brain section filled with these colors, you aren't just seeing cells; you’re seeing a family tree. You’re seeing the history of how a single stem cell populated an entire region of the organ.
It’s worth mentioning that these images are often "false color." The camera on a high-end confocal microscope often captures images in grayscale based on light intensity. The scientist then assigns the colors in software like ImageJ or Zen. So, if a scientist is feeling particularly moody, they might make the nuclei purple instead of the traditional DAPI blue.
The Misleading Nature of "Stock" Stem Cell Photography
We need to talk about the "blue bubble" problem. If you go to a stock photo site and search for stem cells, you’ll see thousands of 3D-rendered, glossy blue spheres floating in a void.
They are fake.
Those aren't photos of stem cells; they are digital illustrations. Real cells are messy. They have jagged edges. They have "blebs"—little protrusions that look like they're bubbling over. They are surrounded by debris, extracellular matrix, and other cells. The sanitized, clinical look of stock photography does a disservice to the complexity of biology. It makes stem cell therapy look like a clean, plug-and-play technology, which it definitely isn't yet.
Confocal vs. Scanning Electron Microscopy
There are two main ways we get these high-end shots.
- Confocal Laser Scanning Microscopy: This is the gold standard for those glowing, multi-colored images. It uses a laser to scan the sample point by point. It can take "slices" of a thick tissue sample and stack them together to create a 3D reconstruction. If you see a photo where you can see inside the cell, it's likely confocal.
- Scanning Electron Microscopy (SEM): These photos are usually grayscale and look incredibly textural. You can see the "hairs" (cilia) or the rough surface of the cell membrane. SEM offers insane resolution—way beyond what light can do—but it requires coating the cells in a thin layer of gold or platinum. It kills the cells, obviously, but the resulting photos are breathtakingly detailed, showing the physical architecture of life.
Spotting the Fakes in "Stem Cell Clinic" Marketing
This is where things get a bit sketchy. Many "unregulated" stem cell clinics use beautiful, high-tech photos of stem cells on their websites to sell treatments for everything from autism to bad knees. Often, the images they use are actually of mesenchymal stem cells (MSCs) derived from bone marrow or fat.
In a real photo of MSCs, the cells look long and spindle-like, almost like tiny shards of glass or fibroblasts. If a clinic is showing you photos of perfectly round, embryonic-looking cells but claims to be using your own fat cells, something is off. MSCs are "multipotent," not "pluripotent." They have a different look and a different limit to what they can do.
Always look for the scale bar. A professional scientific photo will always have a small line in the corner, usually labeled "10 µm" or similar. If there’s no scale bar, it’s probably a marketing graphic, not a scientific record.
The Future: Live-Cell Imaging
The next frontier isn't just a photo; it’s a movie. We used to have to "fix" (kill) cells to get a good picture. Now, with developments in lattice light-sheet microscopy, we can take high-speed photos of stem cells while they are moving, dividing, and even "deciding" what to become.
You can watch a stem cell in a zebrafish embryo migrate from one part of the body to another. You can see the mitochondria—the powerhouses—scurrying around inside the cell like busy ants. This isn't just for show. Seeing how these cells move in real-time helps researchers understand why some stem cell transplants "take" and others fail.
Actionable Insights for Researching Stem Cell Imagery
If you are looking for authentic photos of stem cells for a project, a paper, or just out of curiosity, don't just use a generic search engine. You’ll get buried in stock art and AI-generated nonsense.
- Use the Nikon Small World Gallery: This is an annual competition for photomicrography. The "Stem Cell" category features some of the most scientifically accurate and visually stunning images ever captured.
- Check the Protein Atlas: If you want to see where specific proteins live within a stem cell, the Human Protein Atlas has an incredible database of high-resolution immunofluorescence shots.
- Look for the "Fig. 1" in Journals: Go to PubMed or Google Scholar. Look for open-access papers in journals like Nature Communications or Stem Cell Reports. The "Figure 1" is almost always the characterization photo—the proof that the cells are what the scientists say they are.
- Verify the Source: If an image looks too perfect—perfectly symmetrical, neon-bright without any "noise" or grain—check the credit. If it says "Getty Images" or "Shutterstock," treat it as a representation, not a reality. If it credits a university lab (like the Lanza Lab or the Salk Institute), it’s the real deal.
When you look at a real photo of a stem cell, you’re looking at a biological "blank slate." It’s a cell that hasn't committed to a "career" yet. It could become a beating heart cell, a neuron firing a thought, or a skin cell protecting you from the sun. The photo is just a snapshot of that indecision. Understanding the tech and the "makeup" applied to these cells doesn't make them less amazing; it actually makes the science behind them feel a lot more grounded and real.